Flourish Research - Chicago (Ravenswood)
End Stage Kidney Disease Self-Scheduler
Qualified Participants Must:
- Be 18 to 55 years old
- Be diagnosed with End Stage Kidney Disease
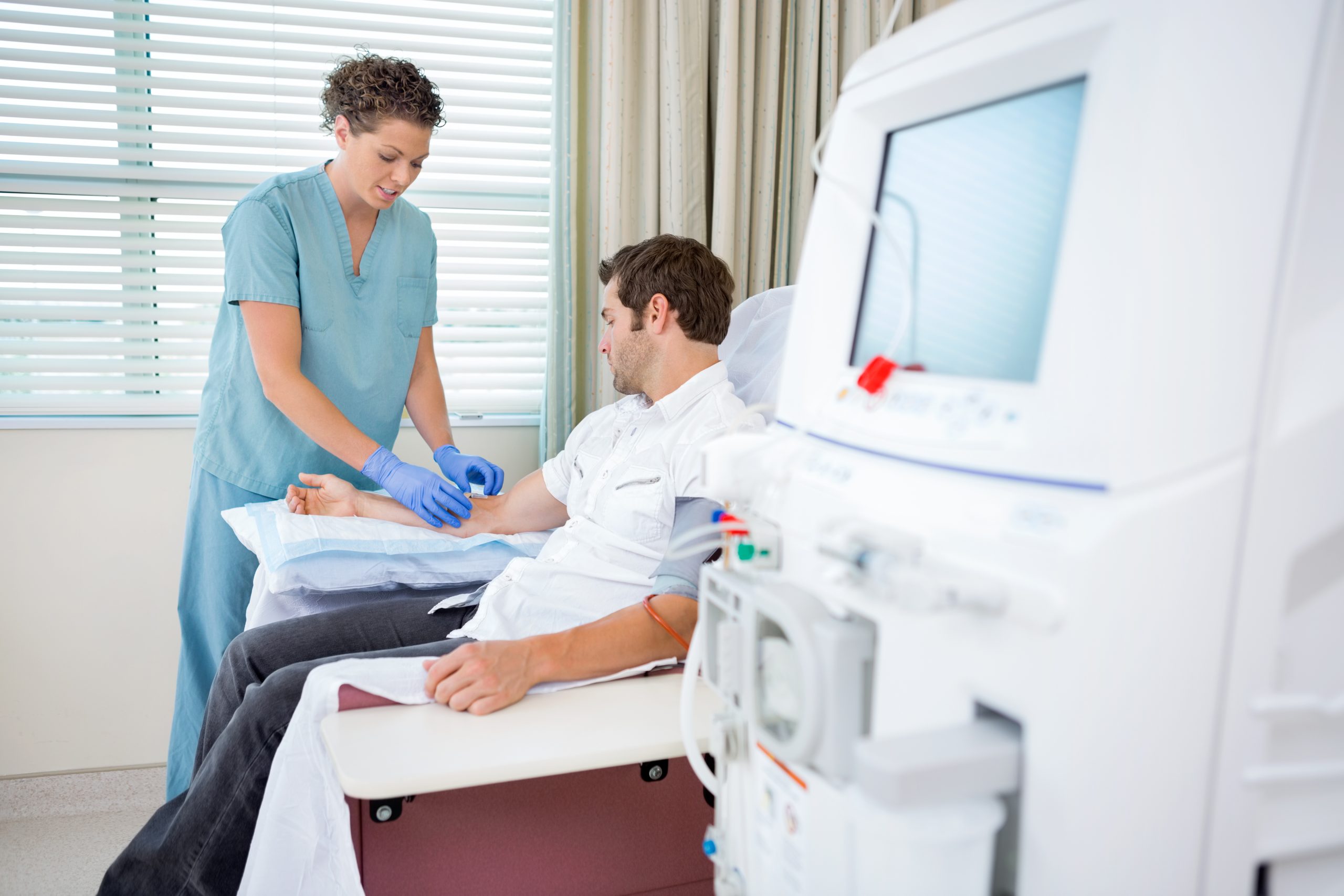
- Have been receiving regular dialysis treatments for at least 3 months
- Be using a tunneled central venous catheter (CVC) for dialysis access
Please note that additional criteria will apply.
Answers Must Be No:
- If female of childbearing potential, are you breastfeeding, pregnant, or planning on becoming pregnant?
- Are you currently taking long-term antibiotics that work against Staphylococcus aureus (S. aureus), such as doxycycline, clindamycin, or trimethoprim-sulfamethoxazole (Bactrim®)?
- Have you ever had an allergic reaction, infusion-related problem, or serious side effect after receiving a monoclonal antibody (mAb) treatment?
- Have you had a heart attack, serious heart problem (acute coronary syndrome), stroke, seizure, or a blood clot (such as deep vein thrombosis or a pulmonary embolism) in the last 3 months? (This does not include clots related to dialysis access)
- Do you have cancer and are undergoing chemotherapy?
- Do you have a scheduled date for a kidney transplant from a living donor?
- Have you had a known Staphylococcus aureus (S aureus) infection in the past 3 months?
- Do you have plans to switch from dialysis to peritoneal dialysis in the next 6 months (about 181 days)?
- Do you have a scheduled date to switch to an arteriovenous (AV) graft for dialysis, or do you already have an AV graft that is in place and still maturing (not yet being used)?
- Do you have a scheduled date to switch to an arteriovenous (AV) fistula for dialysis, or do you already have an AV fistula in place that is maturing, and expected to be used within the next 3 months?
- Do you have a history of injected substance abuse?
- Have you received suvratoxumab in the last 15 months?
- Have you received an investigational drug or device in the last 3 months?
- Do you have HIV, Hepatitis A, or Hepatitis B?
Answers Must Be Yes
- Are you receiving dialysis through a long-term central line (a tunneled catheter) as the main method of access for dialysis?
- If female and sexually active with a fertile male, are you willing to use a highly effective form of contraception throughout the study?
PLEASE NOTE: Please confirm your eligibility before your appointment by calling 312-931-8883
- If you do not see an available appointment, please call 312-931-8883 for availability.
- Transportation may be available. Call 312-931-8883. Compensation may be available for time and travel.
By signing up, you agree to receive text messages and emails about this and similar studies near you. You can unsubscribe at any time. Text messages and data rates may apply.
Schedule Appointment
Flourish Research News
Stay current with the latest clinical trial news and learn how Flourish Research is supporting the advancement of life-changing therapeutics.