Flourish Research - San Antonio
Study Details
Enrolling Clinical Research Studies
Heart Disease Study
Basic Criteria
- Age 18 or older
- Must meet one of the following criteria:
- Have a history of a cardiovascular event such as heart attack, stable or unstable angina, coronary or other arterial revascularization, stroke, or peripheral artery disease. OR
- Be at risk for a first heart condition with one or more of the following conditions: atherosclerotic vascular disease, diabetes, high blood pressure, cigarette smoking (tobacco products and includes vaping), chronic kidney disease (moderate to severe stages), or obesity
- Must be on a high dose statin-atorvastatin 40 mg or rosuvastatin 20 mg-
- Except patients with documented intolerable side effects to at least 2 different statins.
Please note additional criteria may apply

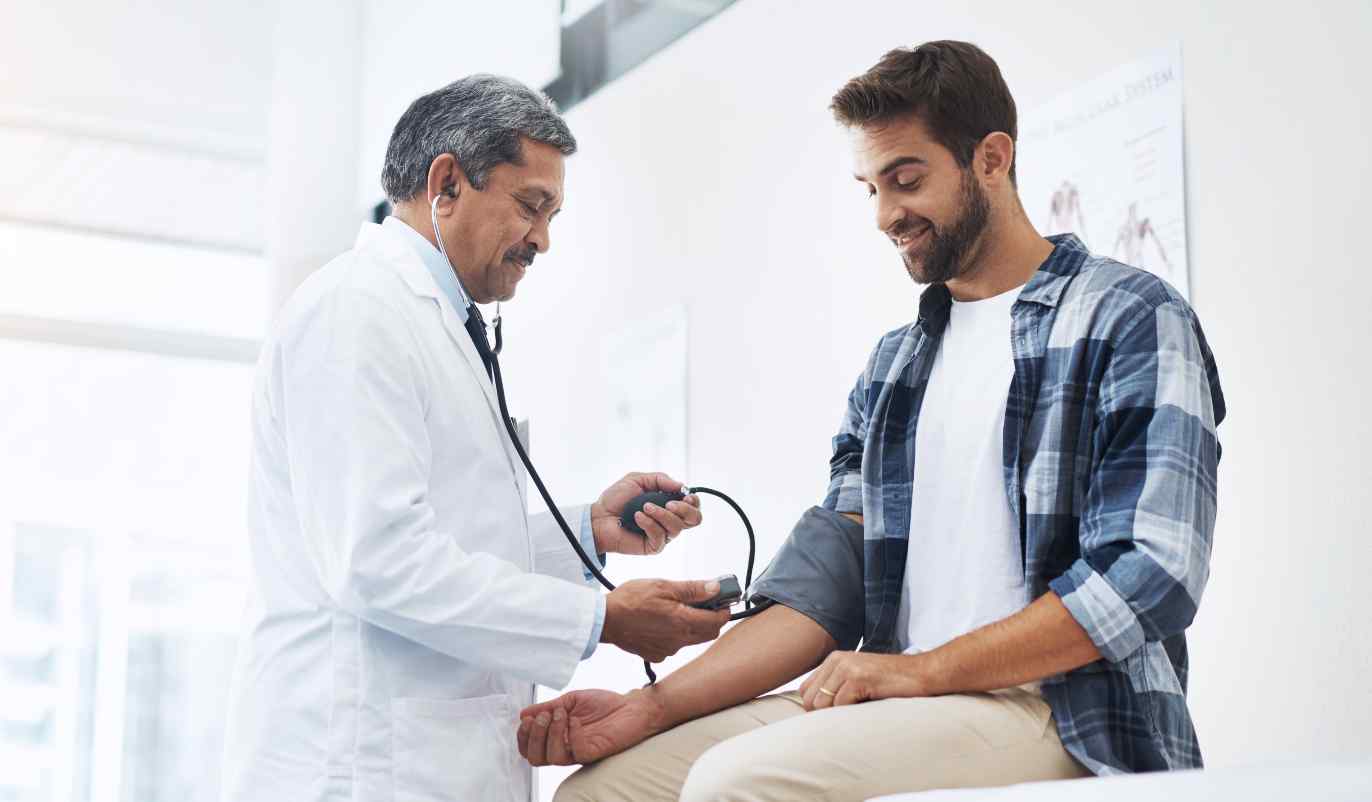

Your Study Experience
We strive to give you the best possible study experience from start to finish!
01
Talk to Our Team
Review your options and decide what is right for you.
02
First Visit
Our team will take the time to ensure you are fully informed about clinical trial treatments, procedures, timelines, compensation, and more. We will check your vitals, review your medical history with you, and do tests such as labs as needed to make sure the study is a good fit.
03
Voluntary Participation
You can stop participating at any time and for any reason.
Hear From Our Participants
Frequently Asked Questions
What are the benefits of participating in a clinical trial?
- Help others and yourself
- Receive study-related care at no cost
- Contribute to advancing medicine
- Get paid to participate
Will I be paid?
- Trials offer compensation as well as additional travel reimbursement in some cases
- You will receive study-related care and tests at no cost
- You do not need to have medical insurance to participate
Flourish Research News
Stay current with the latest clinical trial news and learn how Flourish Research is supporting the advancement of life-changing therapeutics.